By Matthew Littlefield, President & Principal Analyst at LNS Research
It continues to be a common belief that the sooner you identify and correct a quality non-conformance, the less of a negative impact it will have. Within the past decade, though, advancements to quality management software and other enterprise technologies have really taken this idea to new heights and made it significantly more actionable.
Specifically, as next-generation solutions continue to make their way up and down the value chain—from sourcing and design up through manufacturing, distribution, and service—market leaders are working to integrate key data sources and systems to develop closed-loop quality processes, or feedback loops, between critical processes or functional areas.
These closed-loop processes facilitate improvements earlier in the value chain, helping to continuously correct and prevent quality issues while driving costs down. At LNS Research, we’re seeing a lot of interest in this topic, and in this post, we’ll catch you up on what you need to know about closed-loop quality processes. We’ll also share some benchmark data to show why you should care.
What is Closed-Loop Quality Management?
Closed-loop quality management essentially means connecting quality process or performance data from one area to another, always with the goal of improving quality earlier. This connection may be within the same area of the value chain—i.e. just the manufacturing environment—or between different areas of the value chain—i.e. between manufacturing and supply chain management.
To provide perspective, consider the following examples of where companies fail to close the loop:
Closed-loop quality within the manufacturing environment: Every manufacturing organization has some form of corrective and preventive action (CAPA) process for non-conformances as well as an auditing process for ensuring things like quality specifications and training requirements are being met. Unfortunately, many companies don’t effectively capture all of the non-conformances identified in manufacturing into a centralized system. In many cases we’ve heard reports from companies where the warranty team is finding errors and conducting investigations, only to find out later that manufacturing already found and eliminated the root cause but never let any other areas of the organization know. By moving to a centralized system for managing non-conformances, these gaps in understanding can be eliminated.
Closed-loop quality for suppliers: Many discrete manufacturing companies rely on certificates of analysis or limited inspections of incoming raw materials and components to ensure supplier quality. By just focusing on the result—supplier defect rates—improvement becomes difficult. Leading companies are taking a more collaborative approach with suppliers—one that ensures suppliers have robust quality processes in place and that the performance of suppliers’ processes is visible. This in turn drives higher supplier quality performance than could ever be achieved by testing alone and consequently results in fewer issues during manufacturing.
Those are just two examples of challenges in quality and how closed-loop processes can help, and many more cases can be made for the connection of process data between different enterprise systems and processes within them.
In the past, developing such feedback loops was resource-intensive, often requiring considerable time and capital resources. Today, however, much of the excitement around closed-loop quality management stems from relatively recent technological advancements that have enabled organization to move quality performance visibility beyond just the four walls of the manufacturing facility.
Why You Should Care about Closed-Loop Quality Management
To better understand the benefits of closed-loop quality management, we can look at benchmark data from our recent quality management survey. Taken by over 400 quality management executives, the survey asked a number of questions about quality challenges, objectives, technologies adopted, methodologies used, performance metrics, and more.
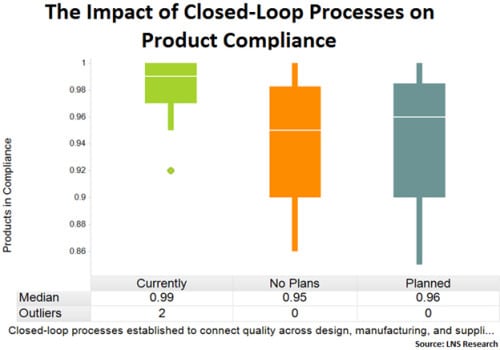
For our purposes, we’ll look at data from two questions. One asked about the rate at which organizations achieved product compliance and the other asked whether or not closed-loop processes were established at the time to connect quality across the value chain. Cross-analyzing this data, the box plot chart below shows a clear difference in performance between those that had closed-loop quality processes established and those that didn’t.
Organizations with closed-loop quality processes established achieved a median product compliance rate of 99%, with a majority of performance ranging from 97-100%. Organizations without the capability experienced significantly more variability in performance, with a majority reported performance ranging from 90% to 98%. This disparity in performance could mean the difference between being first or second to market, avoiding a major product recall, or winning future contracts.
How to Achieve Closed-Loop Quality Management
That’s the what and the why, but the how is a bit more complex. On June 17, I’ll be co-hosting a webinar with Quality Magazine to discuss this very topic. We’ll be covering the importance of closed-loop quality management, and also how market leaders are building such a system today. I’ll be sharing benchmark data as well as best practices across people, processes, and technology for deploying your own closed-loop quality management system. You can sign up by clicking here https://www.qualitydigest.com/content/closed-loop-quality-management-ibt.html.