The highest-performing Manufacturing Execution Systems (MES) reduce the chaotic clutter of paperwork by delivering greater accuracy, precision and quality of online work instructions, while scaling processes across all production centers anytime, anywhere.
Improving manufacturing productivity, quality and compliance by giving operators, supervisors and every plant manager visibility into shop floor operations also differentiates the highest performing manufacturers relying on these systems.
Traits Of High Performance Complex Discrete Manufacturers
They Rely On Manufacturing Execution Systems to bridge the gap between Engineering and ERP Systems
Manufacturers are realizing the need to integrate real-time manufacturing data into their corporate information view. Many production environments have been historically serviced by paper-based processes and homegrown systems that have not kept up with newer requirements for increased speed, agility and traceability.
Manufacturers realize that having disparate, disjointed applications supporting the shop floor increases the difficulty to integrate the required plant data into a complete and accurate top-level view of operations. Higher customer expectations, diverse product lines and complex supply chains are driving plants away from running the facilities with spreadsheets, paper and knowledge held by a few key experienced employees that might be nearing retirement.
An MES manages shop processes and paperwork including (a) track engineering changes, (b) track installation of serialized parts or lot-tracked material, (c) document nonconformance, (d) track component swapping, and (e) document rework and repairs, (f) tooling manufacturing and calibration status.
Production Integrated to Engineering and ERP
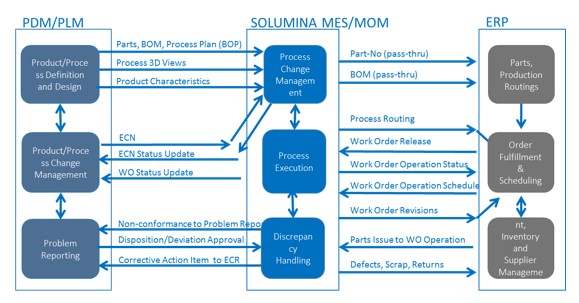
Integration of plant floor systems with engineering and business systems, allows a streamlining of business processes that span across the organization. This includes product and process change management procedures. The discipline and agility acquired through enterprise systems integration can provide significant competitive advantage to an organization.
Many leading manufacturers are integrating their manufacturing system to the Enterprise Resource Planning (ERP), and Product Lifecycle Management (PLM) systems. PLM handles the product definition (including specifications and geometry), ERP handles market facing activities (including planning demand fulfillment, purchasing, and inventory control), and MES handles product realization activities (including work execution, work-in-process tracking, and quality management). The tight integration of these systems can compress time-to-market for both new product and major product upgrades.
Are Exceptional At Product/Process Configuration and Change Management
The manufacturing of a complex product like an aircraft or satellite involves the management of a continuous stream of engineering changes directed at work in process. The integration of the engineering system with MES can create a seamless link between product development, manufacturing planning, and manufacturing execution functions. This link closes the loop on engineering changes, and assure that as-built configurations match as-designed.
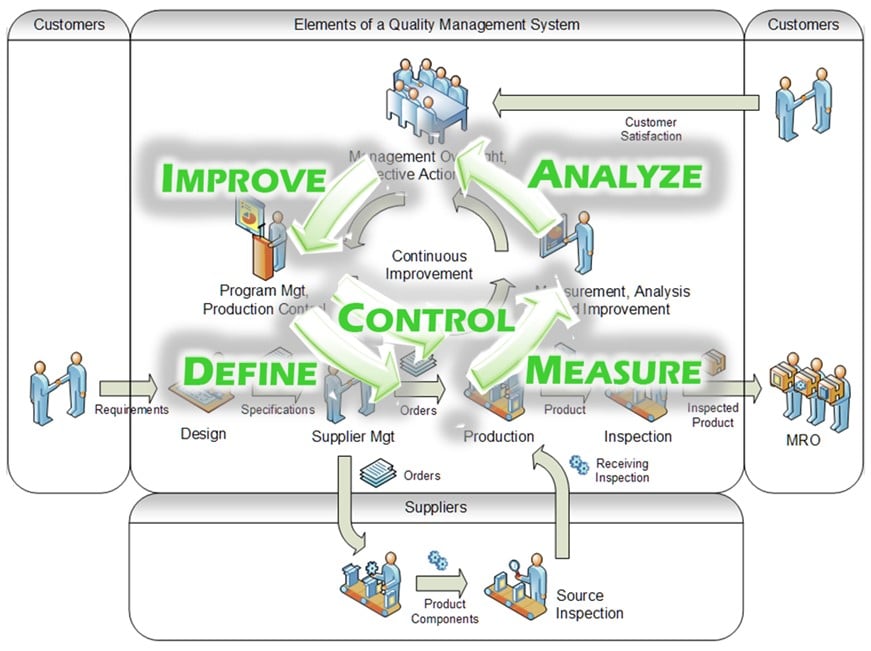
 Integrate and Continually Improve Quality Control Processes
Integrate and Continually Improve Quality Control Processes
Beyond providing visibility into areas for improvements, the manufacturing information system should provide process control procedures to implement and sustain quality improvements. This includes in-process inspection and verification steps, statistical process control (SPC), alerts to out-of-control conditions, and integrated handling for discrepancies found during production (including defect containment and corrective actions) to eliminate recurrence.
With the high investment that goes into these types of products (for parts and labor), they are rarely scrapped. Instead, these industries require rework, repair, and deviation handling procedures to ensure that deviations are documented, reviewed, and approved by the appropriate personnel. The integration of production and quality systems can ensure that deviation instructions cannot be skipped by the mechanic performing the work. Deviation history is also considered part of each product unit history.
Excel At Compliance Management
ISO9001 and its aerospace counterpart, AS9100, establish a standard for a quality management system, or QMS. These standards define the basic practices and procedures organizations must follow to ensure that quality goals are systematically and consistently improved and sustained.
The MES forces adherence to procedures including the following: (a) authoring and revision control of work instructions, (b) the execution sequence of manufacturing orders and data collection, (c) the use of proper resources, and (d) the handling and approval of any deviations during production via the configured business process workflows and change management functions.
Companies must be able to demonstrate adherence to process control and risk management. It’s not just a matter of satisfying compliance — these procedures and processes will also reduce cost, improve product quality, and ultimately improve customer satisfaction and the bottom line.
These and many other traits differentiate high performance manufacturers relying on MES to accelerate and improve compliance, quality management, resource management, product unit history and records archival.